Digital Imaging versus Ophthalmoscopy for congenital cataract screening

Photo courtesy of 42 Technology Limited
Thank you!
Thanks to your participation, nearly 41,000 babies have been included in the DIvO study.
We have learnt a lot about what works well and what doesn’t using the current device prototype, and this will help us to make an even
better device to help babies in the UK and overseas, allowing early detection of cataract and prevention of sight loss.
Findings so far..
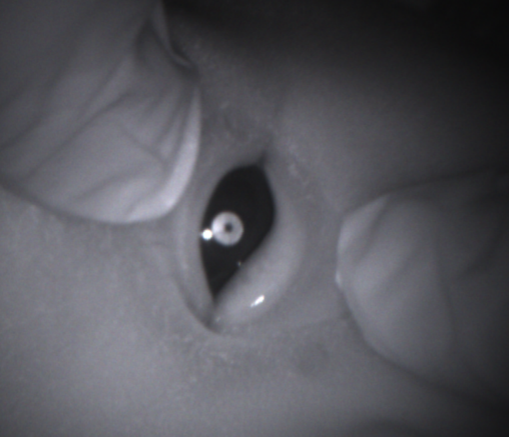
Our data so far shows that several cataracts - like the one shown here - were not detected using the standard screening test,
but were detected with Neocam.
An overwhelming majority of screeners also preferred to use Neocam compared to the standard test.
To be able to fully compare the accuracy of the two methods, we will need to wait until early 2027 when all the babies we recruited have all turned one year old. When we have analysed the results, the study findings will be available here on the website.

Your data
If you have previously enrolled for the study, we will continue to store your contact details
and NHS number in the secure trial system database. This data may be used for analysis but any identifying information
will be removed. Your name, NHS number and email will be stored by Cambridge University Hospitals NHS Trust (CUH) only for the duration
of the study and will not be used for any other purpose. Your details will not be shared with any third parties.
For further information on how CUH protects your data please see the CUH data and privacy information page.
You can decide to withdraw your baby from the study at any time without giving a reason and this will not affect your baby's clinical care in any way.
Any data collected before you inform us of your decision to withdraw, such as the eye photographs or the findings of the newborn eye examination, will continue to be stored securely and used in the study analysis.
To withdraw your baby from the study, please contact the central DIvO study team:
Email: add-tr.divo@nhs.net
Telephone: 01223 254665
Postal Address: The DIvO Study Office, Cambridge University Hospitals NHS Foundation Trust, Addenbrooke's Hospital, Coton House Level 6, Box 401, Hills Road, Cambridge, CB2 0QQ.
